Array by Precision Neuroscience.
Source: Precision Neuroscience
It happened so fast that Craig Mermel missed it.
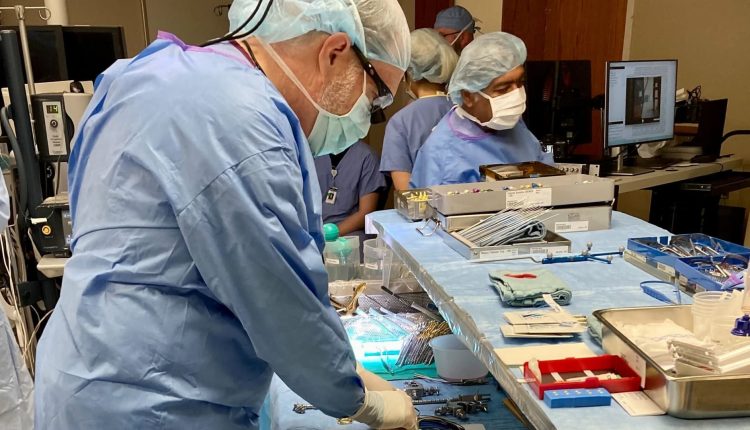
He stood in a busy West Virginia operating room, waiting for a surgeon to insert Precision Neuroscience’s nerve implant system into a conscious patient’s brain for the first time. Mermel, Precision’s president and chief product officer, said he looked away for a moment, and when he turned back, the company’s wafer-thin electrode array was in place.
Within seconds, a high-resolution representation of the patient’s brain activity streamed across a screen in real time. According to Precision, the system had produced the highest resolution picture of human thought ever recorded.
“It was incredibly surreal,” Mermel said in an interview with CNBC. “The nature of the data and our ability to visualize it gave me… goosebumps.”
The procedure observed by Mermel was the company’s first human clinical trial.
Founded in 2021 by a co-founder of Neuralink, Elon Musk’s brain-computer interface startup, Precision is an industry competitor working to help patients with paralysis operate digital devices by decoding their neural signals. A BCI is a system that decodes brain signals and translates them into commands for external technologies. Several companies such as Synchron, Paradromics and Blackrock Neurotech have also developed devices with this ability. Precision announced a $41 million Series B funding round in January.
The company’s flagship BCI system, the Layer 7 Cortical Interface, is an array of electrodes that resembles a piece of duct tape. Because it’s thinner than a human hair, Precision says it can conform to the brain’s surface without damaging tissue. In the study, Precision’s system was temporarily attached to the brains of three patients who had previously undergone neurosurgery to remove tumors.
Because the technology worked as expected, future studies will explore further applications in clinical and behavioral contexts, Mermel said. If the studies go according to Precision’s plan, patients with serious degenerative diseases like ALS could eventually be able to communicate with loved ones again by moving cursors, typing, and even accessing social media with their minds.
Although a human trial is an important milestone, the road to commercialization of this type of technology is long. Precision has yet to receive US Food and Drug Administration approval for its device, and the company must work closely with regulators to successfully complete several rigorous rounds of testing and data security collection.
As of June, no BCI company has succeeded in receiving the FDA’s final seal of approval.
“The goal is to provide a device that can help people with permanent disabilities. So this is the first step,” Mermel said. “Now the real work begins.”
Doctors prepare Precision Neuroscience’s system.
Photo: Anna von Scheling
according to dr Benjamin Rapoport, Precision’s co-founder and chief science officer, has offered to support the company’s pilot clinical study from several different academic medical centers. The company worked with West Virginia University’s Rockefeller Neuroscience Institute, and the two organizations prepared for the procedures more than a year in advance, Rapoport said.
Rapoport, who has been working on the BCI technology for more than 20 years, said it was an “incredibly gratifying” milestone to see the Precision technology on the brain of a human patient for the first time.
“I can’t really describe emotionally what that’s like,” he said. “It was tremendous.”
dr Peter Konrad, Chair of the Department of Neurosurgery at the Rockefeller Neuroscience Institute, was the surgeon who physically placed Precision’s system in patients’ brains during procedures.
Konrad said it was a simple process that felt like placing a piece of tissue paper on the brain.
The patients had the Precision system in their brains for 15 minutes. One of them slept during the procedure, but two patients were woken up so Layer 7 could capture their brain activity while they were speaking.
“I’ve never seen that amount of data, 1,000 channels in real time, electrical activity just sloshing across the brain while someone was talking,” Konrad said in an interview with CNBC. “It was literally like watching someone think. It’s pretty amazing.”
Electrodes are already being used in the field to help neurosurgeons monitor brain activity during a procedure. However, the resolution of conventional systems is low. Konrad said standard electrodes are about 4mm in size, while Precision’s array can fit 500 to 1,000 contacts on that size.
“It’s the difference between looking at the world with an old black and white camera and looking at it in HD,” he said.
Konrad said it is too early for the patients in this study to see the direct benefits of this technology.
Precision Neuroscience’s offer compared to a penny.
Photo: Anna von Scheling
Ultimately, Precision hopes its technology will not require open-brain surgery at all. In an interview with CNBC in January, co-founder and CEO Michael Mager said a surgeon should be able to implant the array by drilling a thin slit in the skull and sliding the device into a mailbox like a letter. The slit would be less than a millimeter thick, so small that patients’ hair would not have to be shaved for the procedure.
Precision’s minimally invasive approach is intentional, as competing BCI companies such as Paradromics and Neuralink have developed systems designed to be inserted directly into brain tissue.
Rapoport said inserting a BCI into the brain would provide a clear picture of what each neuron is doing, but it carries the risk of tissue damage and is difficult to scale up. He said the level of detail isn’t required to decode speech or achieve the other features Precision is aiming for, so it’s a compromise the company is ultimately willing to make.
In the coming weeks, Precision will perform the same procedure on two additional patients as part of its pilot clinical study. Rapoport said the company has submitted its initial results to a scientific journal and that publishing the data is a “big next step.”
Precision is also working on similar studies with healthcare systems like Mount Sinai in New York City and Massachusetts General Hospital in Boston, and Rapoport said Precision hopes to get full FDA clearance for its first-generation device within the next year.
“We are extremely pleased with the initial results,” said Rapoport. “If you’re lucky, there are a few times in your life that you see something before anyone else in the world sees it.”

Comments are closed.