A federal judge in Texas publicly announced Monday afternoon that he had scheduled a hearing in a case seeking to overturn the Food and Drug Administration’s approval of the abortion pill mifepristone after media criticism of him for trying to keep the procedure secret until the last minute.
Judge Matthew Kacsmaryk of the US Northern District of Texas ordered hearings in the case to be held Wednesday at 9 a.m. CT, according to a court filing. The hearing will be held in Amarillo, Texas.
Kacsmaryk was appointed by ex-President Donald Trump.
The Washington Post reported over the weekend, citing people familiar with the matter, that Kacsmaryk set the hearing date during a Friday conference call with attorneys involved in the case, but didn’t want to announce the date until late Tuesday.
Media submitted a letter on Monday urging Kacsmaryk to announce the date of the hearing immediately. Outlets have included NBCUniversal News Group, which owns CNBC, the Washington Post, ProPublica, the Texas Press Association, and Gannett, among others.
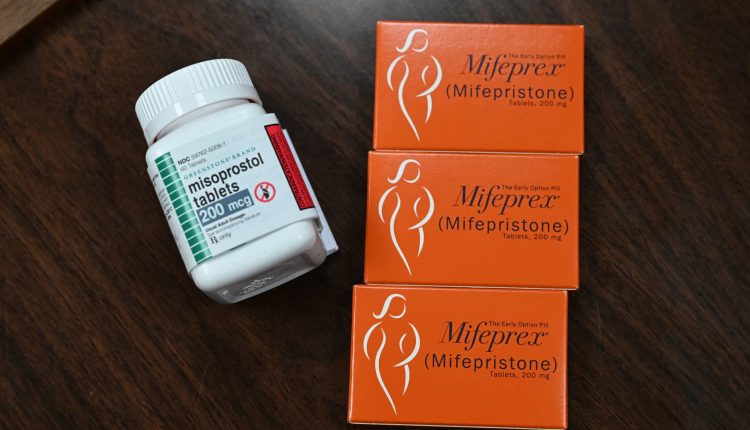
A coalition of anti-abortion doctors urged Kacsmaryk in November to order the FDA to withdraw approval for mifepristone, which has been on the US market since 2000. They argued that the way the FDA approved mifepristone violated federal law.
Join CNBC’s Healthy Returns on March 29, where we’re hosting a virtual gathering of healthcare CEOs, scientists, investors and innovators to reflect on the advances made today to reinvent the future of medicine. We also have an exclusive look at the best investment opportunities in biopharma, healthcare technology and managed care. Learn more and register today: http://bit.ly/3DUNbRo
The FDA has firmly denied the group’s claims, warning that withdrawing mifepristone from the US market would put women’s health at risk and dramatically affect public interest. The agency’s attorneys said that rescinding the approval would also weaken the FDA’s authority.
“If long-standing FDA drug approvals were so easily waived decades after they were granted, pharmaceutical companies could not confidently rely on FDA approval decisions to develop the pharmaceutical drug infrastructure Americans depend on to treat a variety of health conditions,” he said the attorneys for the Biden administration wrote.
Mifepristone, used in combination with misoprostol, is the most common method of terminating a pregnancy in the United States, accounting for about half of all abortions.
Disclosure: NBCUniversal is the parent company of CNBC.

Comments are closed.