The invention of “knock-on chemistry” opens up new frontiers in response dynamics – Watts Up With That?
New experimental findings cast doubt on the 90-year-old theoretical model of the transition state in chemical reactions
UNIVERSITY OF TORONTO
Research news
IMAGE: THE DESIGN OF THE ENERGY BARRIER BY AN ARTIST THAT A REAGENT FLUORINE ATOME MUST CROSS ON ITS PATH TO CONSTITUTE A PRODUCT AS A RESULT OF THE FLUOROMETHYL MOLECULE TO CROSS ITS RESULTS: ILLY CREDIT: ILLY CREDIT: … Show more
TORONTO, ON – Research by a team of chemists at the University of Toronto, led by Nobel Prize winner John Polanyi, sheds new light on the behavior of molecules that collide and exchange atoms during chemical reactions. The discovery casts doubt on a 90-year-old theoretical model of the behavior of the “transition state” between reagents and products in chemical reactions, thus opening up a new area of research.
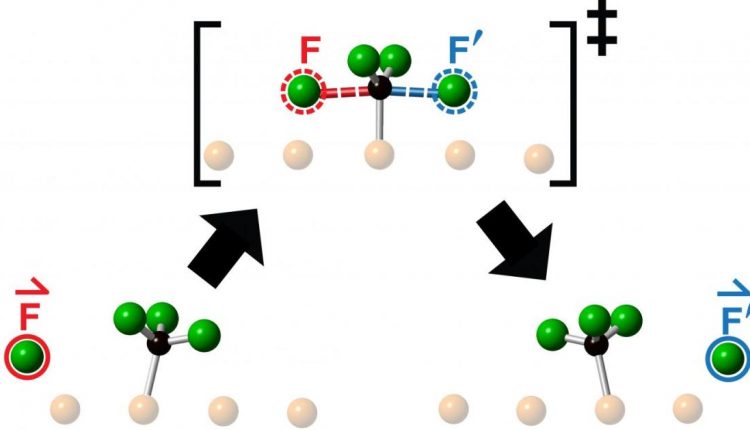
The researchers studied collisions obtained by launching a fluorine atom in the center of a fluoromethyl molecule – made up of a carbon atom and three fluorine atoms – and observed the resulting reaction using scanning tunneling microscopy. What they saw after each collision was the ejection of a new fluorine atom, moving collinearly along the continuation of the approaching direction of the incoming fluorine atom.
“Chemists are constantly throwing molecules at other molecules to see what is happening or hoping to create something new,” says Polanyi, a professor in the Department of Chemistry in the Department of Arts and Sciences at T University and lead author of a study published this month in Communications Chemistry. “We found that aiming a reagent molecule at the center of a target molecule limits the movement of the resulting product to a single line, as if the product had been hit directly. The surprising observation that the reaction product emerges in a straight line and moves in the same direction as the incoming reagent atom suggests that the movements that lead to the reaction resemble a simple further transmission of the momentum.
“The preservation of linear momentum that we observe here suggests a short-lived“ transition state ”rather than the previous view that there is enough time for motion to be randomized. I think Newton would have been happy that nature allowed a simple event to describe something as complex as a chemical reaction, ”says Polanyi.
The team, which included senior research fellow Lydie Leung, graduate student Matthew Timm, and graduate student Kelvin Anggara, had previously determined the means to control whether a molecule aimed at another collided either head-on with its target or around a specific one Amount Missed – a amount known as the strike parameter. The higher the impact parameter, the greater the distance by which the arriving molecule will miss the target molecule. For the new work, the researchers used a zero impact parameter to achieve a head-on collision.
“We call this new type of one-dimensional chemical reaction ‘knock-on’ because we find that the product is switched on along the continuation of the reagent approach,” says Polanyi. “The movements are similar to the knocking of steel balls in a Newtonian cradle. The cradle’s steel balls do not jumble, but instead efficiently transmit momentum along a single line.
“In a similar way, our kick-off reactions transfer energy along rows of molecules, which promotes a chain reaction. This conservation of reaction energy in chemistry could be useful as the world moves towards energy conservation. At the moment it serves as an example of a chemical reaction in its simplest form. “
It has been known for well over a century that there is an energy barrier that chemical reagents must cross on their way to form reaction products. In a critical configuration, there is an activated transition state shortly on top of the barrier – no transition state, no reaction.
According to Polanyi, observing the collinear “impact” provides insight into the reactive collision complex, which lasts about a million-millionth of a second. “Our results clearly show that the transition state at the top of the energy barrier lasts so little time that it cannot completely encode its impulses. Instead, it remembers the direction the attacking fluorine atom came from. “
In the 1930s, chemists began to calculate the likelihood of a transition state forming assuming that it encoded its energy like a hot molecule. While this was an assumption, it appeared to be well established and led to statistical “transition state theory” of reaction rates. This is still the preferred method for calculating reaction rates.
“With the ability to observe the reagents and products at the molecular level, you can see exactly how the reagents approach and how the products then separate,” says Polanyi. “However, this contradicts the classic 90-year-old statistical model. If the energy and momentum were randomized in the hot transition state, the products would have no clear memory of the direction of approach of the reagents. Energy randomization would work to erase this memory. “
The researchers say that the observed directional movement of the reaction products favors a deterministic transition state model to replace the longstanding statistical model. In addition, the observed reaction dynamics allow the reagent energy to be passed on in successive collinear collisions.
###
The research was funded in part by the Canadian Science and Engineering Research Council (NSERC) and the University of Toronto’s NSERC General Research Fund. Theoretical calculations were carried out on the Niagara cluster of the SciNet HPC Consortium.
From EurekAlert!
Like this:
Loading…

Comments are closed.